 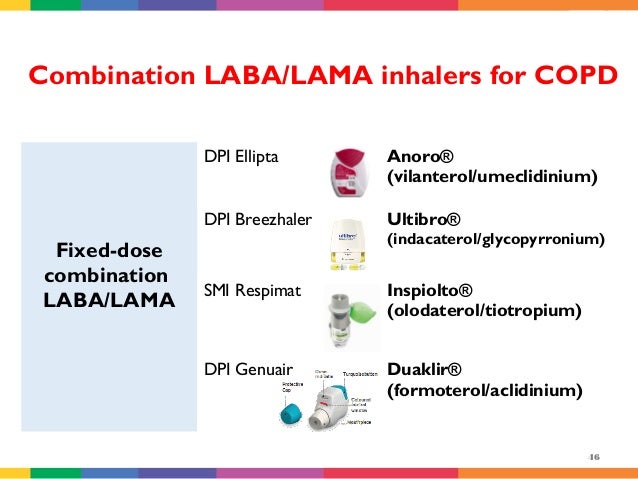
More recently, in 2020, the drug was approved to treat asthma. In 2017, the Food and Drug Administration (FDA) approved Trelegy Ellipta to treat COPD. Effectivenessįor information about the effectiveness of Trelegy Ellipta, see the “ Trelegy Ellipta for COPD” and “ Trelegy Ellipta for asthma” sections below. Trelegy Ellipta is approved for use in adults ages 18 years and older. This medication is taken as one inhalation (puff) of Trelegy Ellipta once each day. The word “Ellipta” refers to the specific type of dry powder inhaler that contains the drug. “Trelegy” is the name of the medication inside the inhaler. vilanterol, which is a long-acting beta2-agonist (LABA).umeclidinium, which is an anticholinergic.fluticasone, which is an inhaled corticosteroid (ICS).It contains these three active drugs that help relieve breathing symptoms of COPD and asthma: Trelegy Ellipta is a combination medication. For more information about Trelegy Ellipta’s approved uses, see the “ Trelegy Ellipta for COPD” and “ Trelegy Ellipta for asthma” sections below. However, Trelegy Ellipta isn’t approved for use as a rescue inhaler to ease sudden breathing problems caused by either COPD or asthma. chronic obstructive pulmonary disease (COPD), which is a group of lung conditions that include emphysema and chronic bronchitis.It’s FDA-approved for the long-term maintenance treatment of: For further information see text next to the product.Trelegy Ellipta is a brand-name prescription medication. Products shown as blue have multiple indications with varying formulary classifications. 
Treatment should be supplied by the hospital for the duration of the treatment course. Treatment to be initiated in hospital prior to handover. Treatment may be initiated in Primary Care on the recommendation of a consultant/specialist. Treatment may be initiated in either Primary or Secondary Care.Īvailable for restricted use under specialist supervision. Treatment may be initiated in either Primary or Secondary Care, including Community Pharmacies for minor illnesses included in the NHS Pharmacy First Scotland Approved List. Trixeo ® Aerosphere ® (formoterol fumarate dihydrate 5micrograms/ glycopyrronium 7.2micrograms/ budesonide 160micrograms) is not routinely available as there is a local preference for alternative medicines. after dispensing, in-use shelf-life 3 months at a temperature up to 25☌.Is licensed for maintenance treatment of asthma, in adults 18 years and over who are not adequately controlled with a maintenance combination of a long-acting beta2-agonist and high dose of inhaled corticosteroid, and who experienced one or more asthma exacerbations in the previous year Pressurised inhalation, solution: beclometasone dipropionate 172micrograms/ formoterol fumarate dihydrate 5micrograms/ glycopyrronium 9micrograms: after dispensing, in-use shelf-life 4 months at a temperature up to 25☌.prior to dispensing it must be stored in a refrigerator (2☌ - 8☌).Pressurised inhalation, solution: beclometasone dipropionate 87micrograms/ formoterol fumarate dihydrate 5micrograms/ glycopyrronium 9micrograms: should be used within 6 weeks after first opening the pouch.the recommended and maximum dose is two inhalations twice daily.Trimbow ® NEXThaler ® (DPI) 88/5/9 (medium dose ICS)ĭry powder inhaler: beclometasone dipropionate 88micrograms/ formoterol fumarate dihydrate 5micrograms/ glycopyrronium 9micrograms: if stored in a refrigerator allow the inhaler to return to room temperature for at least an hour before use.must be used within six weeks of first opening the tray.the recommended and maximum dose one dose once daily.Trelegy ® Ellipta ® (DPI) (medium dose ICS)ĭry powder inhaler: fluticasone furoate 92micrograms/ umeclidinium 55micrograms/ vilanterol 22micrograms: Triple Therapy products (ICS + LABA + LAMA) NICE TA138: Asthma (in adults) - corticosteroids NICE TA131: Asthma (in children) - corticosteroids NICE NG115: Chronic obstructive pulmonary disease in over 16s: diagnosis and management SIGN 158: British guideline on the management of asthma Formulary Chapter 3: Respiratory system - Full Chapter 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
